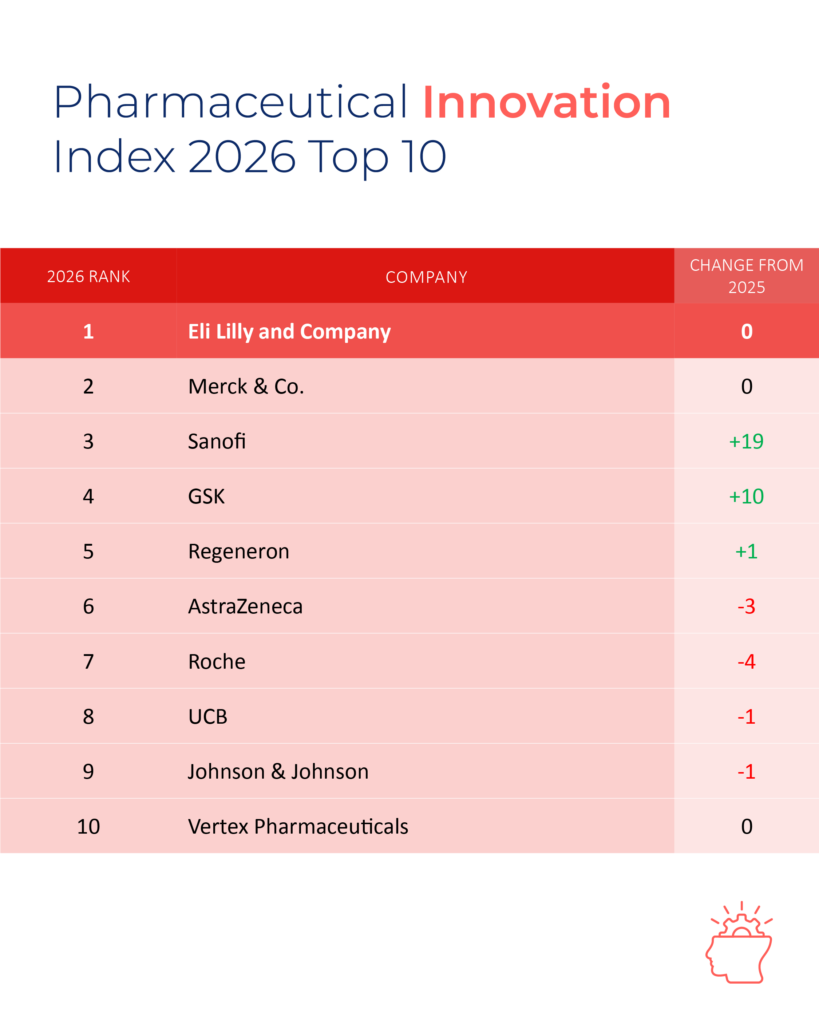
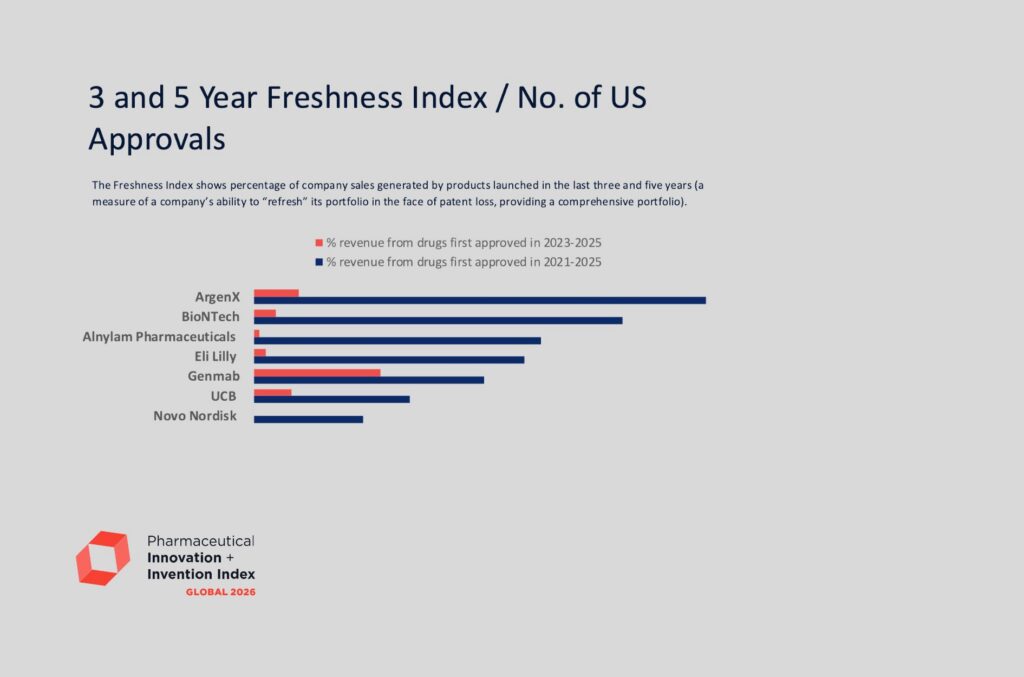
Eli Lilly top 2026 Global Pharmaceutical Innovation & Invention Index
Eli Lilly First Ever Company to Top Both Rankings in the 2026 Pharmaceutical Innovation and Invention Index See the full results at https://www.ideapharma.com/pii/report/global/ LONDON, UK — May 19th 2026 — The 2026 Global Pharmaceutical Innovation […]